Taking a fourth-to-market product straight to the top and keeping the momentum going.
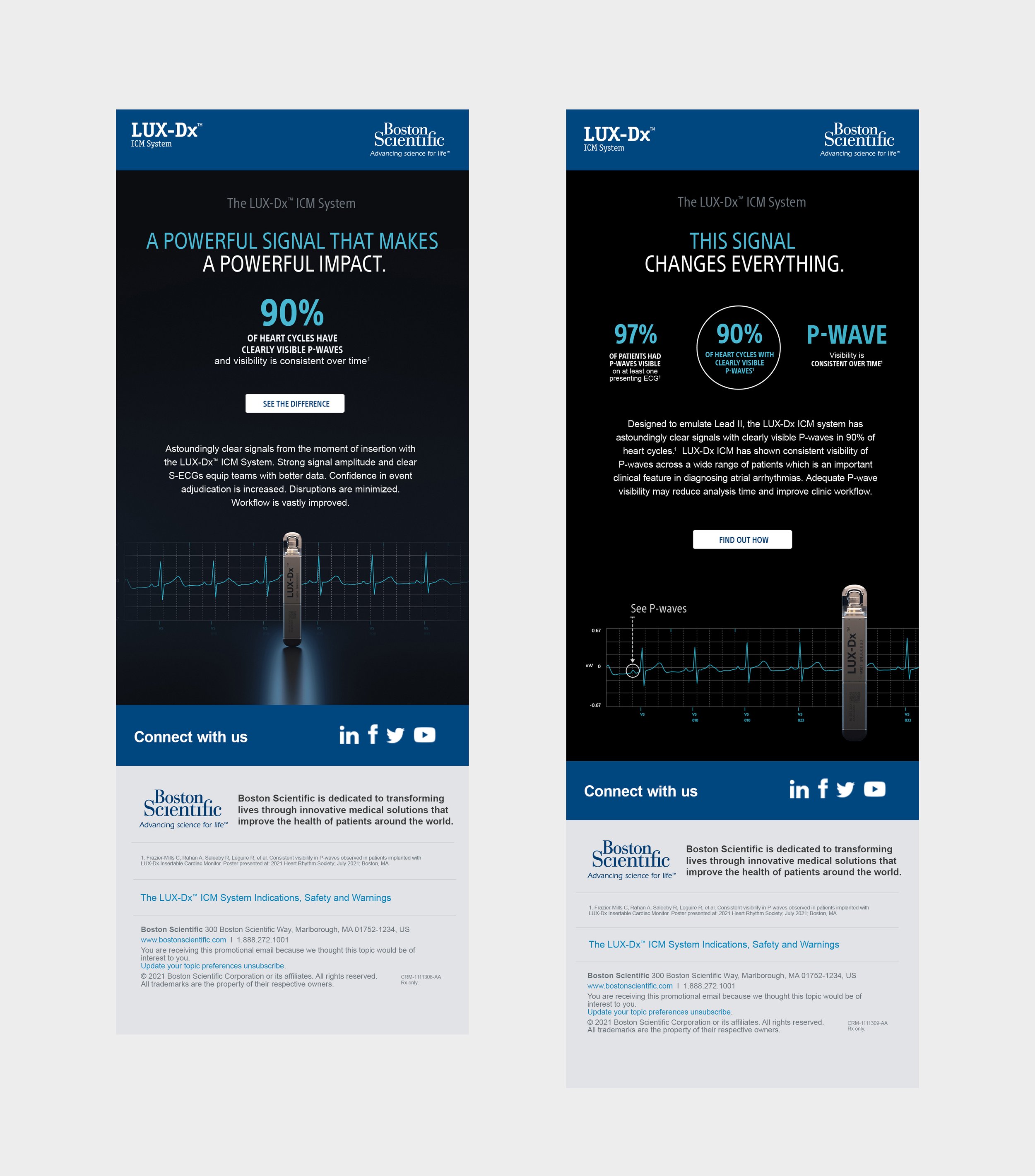
Current ICMs (Insertable Cardiac Monitors) had a bad reputation. Notorious for data burden and a propensity for false positives they were responsible for lost time, frustrated teams and worried patients. Boston Scientific’s new insertable cardiac monitor, the LUX-Dx™ had two differentiating features but no human data or verified statistical claims at the time of launch in order to challenge Medtronic, Abbott and Biotronik.
And, Boston Scientific wanted to gain share quickly.

With a more than likely superior device but no substantiating claims, how do we say we are better … without saying we are better?



Knowing the current frustration in the market and appetite for something better, we leaned into existing pain points. Creating a campaign using FOMO to paint a picture of what an ICM could be — positioning Boston Scientific’s ICM as a device implanters simply couldn’t resist trying.





Initial launch results for the LUX-Dx ICM surpassed projected expectations. Now with new found data and impressive implant feedback there were opportunities to share these results based on data and continue to win share shift and gain adoption.